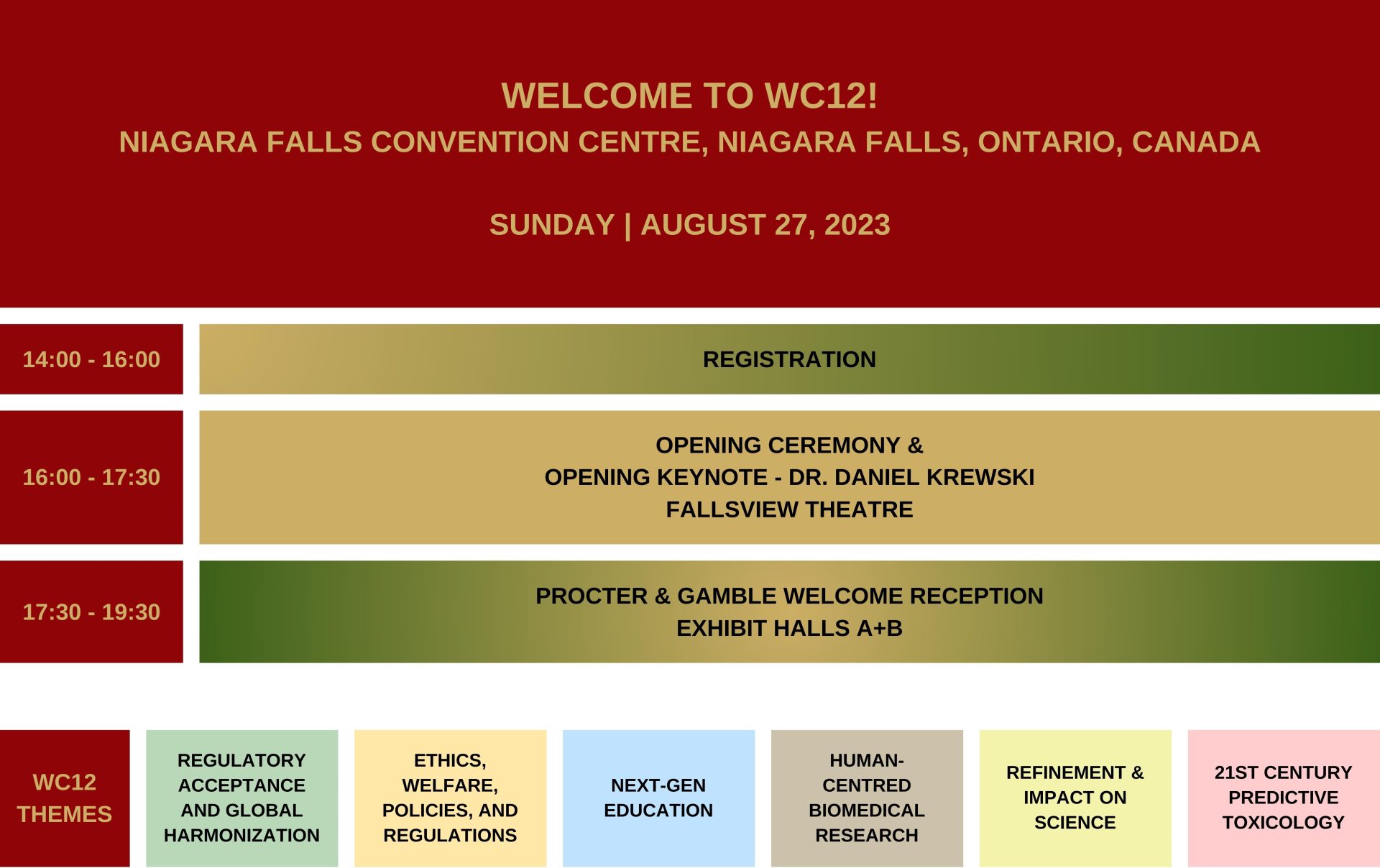
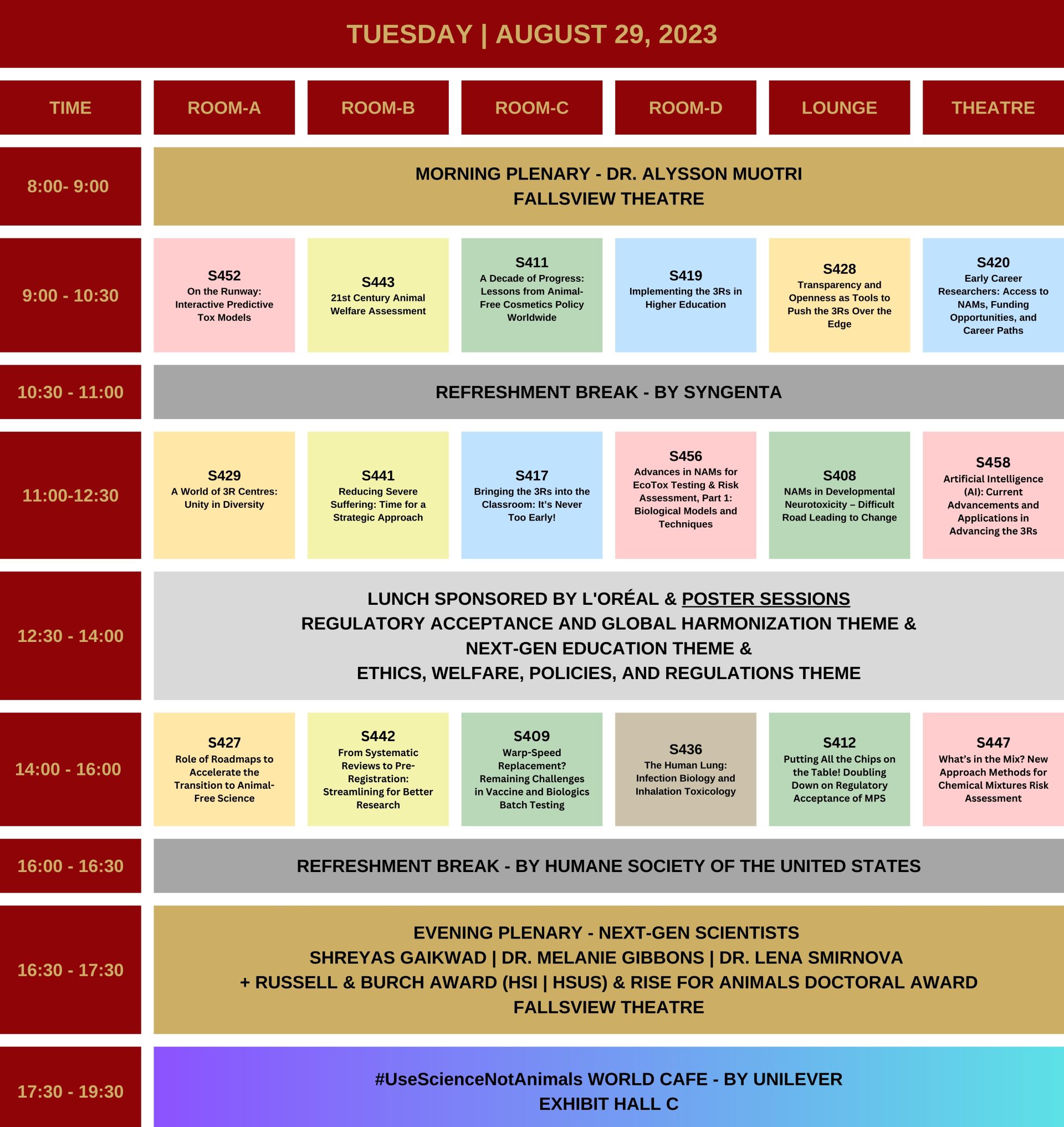
Program-at-a-glance
First draft of full program now available.
6 Parallel Tracks.
60 Platform Sessions.
400+ Speakers.
400+ Poster Sessions.
Bucket-list Socials.

3Rs OVER THE EDGE
REGULATORY ACCEPTANCE & NEXT-GEN EDUCATION
REGULATORY ACCEPTANCE
Over the past decade, the field has exploded with innovative new approach methodologies (NAMs) poised to reduce and replace animal testing; however, their global acceptance into the regulatory risk assessment process has been limited to specific assays and only in certain countries. To fully realize the 21st-century vision, it is critically important to capture the great potential of NAMs and incorporate them into safety & risk assessment pipelines globally. What better place to address this pressing issue of our time than at a multi-stakeholder setting at WC12?
NEXT-GEN EDUCATION
Alongside regulatory acceptance comes the paramount responsibility of educating the next generation to carry on the ground-breaking advances currently being made in this field. We need these next-gen scientists to be on grant review panels to defend NAMs; to be journal editors and reviewers to accept manuscript based solely on human data; to conduct only animal free research; and to be regulators to make crucial decisions on human health risks using NAMs. (Photo here of trainees at the Canadian Centre for Alternatives to Animal Methods).





It's not just scientific sessions!
To spur progress and advance a field, an interconnencted community is key. In keeping with the traditions of the iconic World Congress series, we are planning an exciting social program to reconnect with each other in-person after six years!
Reconnect. Rejuvenate. Return Inspired.
Please click on the button below for an overview of satellite events happening during WC12.








